 They have the optimal number of electrons helium has to Neon has 10. Now we said that all these elements of the periodic table are all trying to become just like our noble gasses are Group eight a elements. And here, if we're dealing with a neutral element, it'll have the same number of electrons. So beryllium a year has an atomic number four, which means it has four protons. For a neutral element, it's number off electrons is equal to its number of protons. For example, we'd say that beryllium has an atomic number four, so it has four protons. Elements now recall that the atomic number, which uses the variables e of an atom, equals the number of protons within its nucleus. We'll talk about those later on the four. Remember your main group elements are the elements found in Group One A or Group one, two way or group to and then from three, a tow eight A or what we call groups 13 to 18. So let's talk about first our main group elements. Where we go into, how many electrons do they gain? How many electrons do they lose and in that way, know what type of ions are possible for certain types of elements. 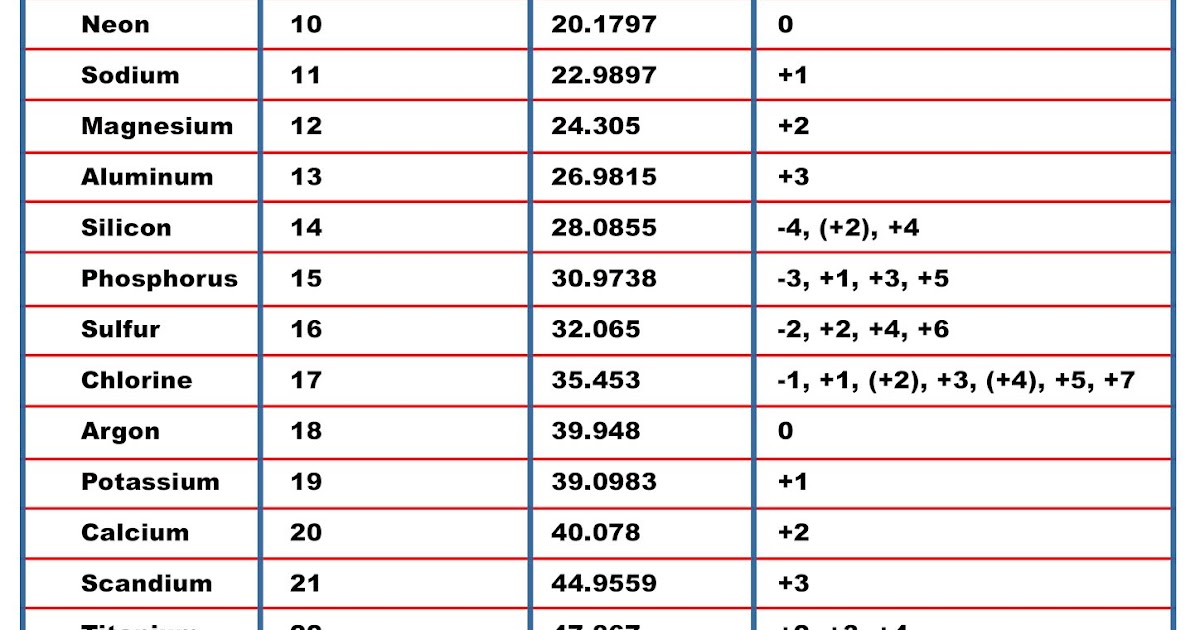
Okay, now that we know this fundamental idea, let's move on to our next video. Non metals tend to gain electrons to become negatively charged. So just remember, remember, metals tend to lose electrons to become positively charged. Chlorine is a non metal, so it makes sense it could gain electrons to become negatively charged. So This would be our answer then, if we look at the last one. Medals tend to lose electrons to become positively charged, so it doesn't make sense that l would have a negative three charge here, So this is unlikely. Medals tend to lose electrons to become positively charged, so that positive charge is a possibility. So this is a likely set up for R B I on next oxygen o oxygen is a non metal nonmetal tend to gain electrons have become negatively charged. We know that our B has is a metal and therefore it makes sense that it would have a positive charge. So if we take a look here, we have our be looking on a periodic table. And remember, non metals tend to gain electrons and therefore becoming negatively charged. They're going to become positively charged. So before we look at this question, remember, we're gonna say that medals, they tend to lose electrons, so they're gonna lose electrons. It says, from what you know about ion formation in the periodic table, which ion would be unlikely to occur. In the next video, we'll see exactly how many electrons will we lose or gain for any particular element. So just remember, the whole reason elements gain and lose electrons is to become just like the noble gasses. I'm gaining negatively charged electrons, so I'm gonna become more negative as a result. So non metals tend to gain electrons to become negatively charged ions called an ions. If medals lose electrons, then non metals must be gaining electrons. Medals will go in greater detail on what types of metals are Type one medals and what other types of metals are. When metals have multiple charges, then they're referred to as type two. When a metal possesses one charge, we refer to them as type one medals. AT T could represent the positive charge that the ion gains, and it makes sense because if you're losing negatively charged subatomic particles in the form of electrons, you're going to become mawr positively charged Now, medals themselves medals can have either one type of positive charge or multiple types of positive charges. When it comes to medals, medals tend to lose electrons to become positively charged ions called cat ions. So if there number of electron matches one of the nearest noble gasses, now we're going to stay here. But just realize when it comes to the elements, the leader lose or gain electrons. What that means specifically will talk about several chapters from now. 
The reason these elements want to become the noble gas is because noble gasses themselves have the optimal number off outer shell electrons. Okay, so remember two different ways to describe the same column of the periodic table. Now, remember, your noble gasses are the elements that are found in Group eight A or Group 18. Image showing periodicity of the chemical elements for molar volume in a 3D periodic table column style.Now the elements of the periodic table will either lose or gain electrons to become just like the noble gasses. Image showing periodicity of the chemical elements for molar volume in a 3D spiral periodic table column style. Image showing periodicity of the chemical elements for molar volume in a spiral periodic table heatscape style. 
Image showing periodicity of the chemical elements for molar volume in a periodic table heatscape style. Image showing periodicity of the chemical elements for molar volume in a periodic table cityscape style. The molar volume is equal to the atomic weight divided by the density. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
